In this edition of the FNA Deep Dive, FoodNavigator-Asia focuses specifically on the regulatory landscape for this sector to find out more.
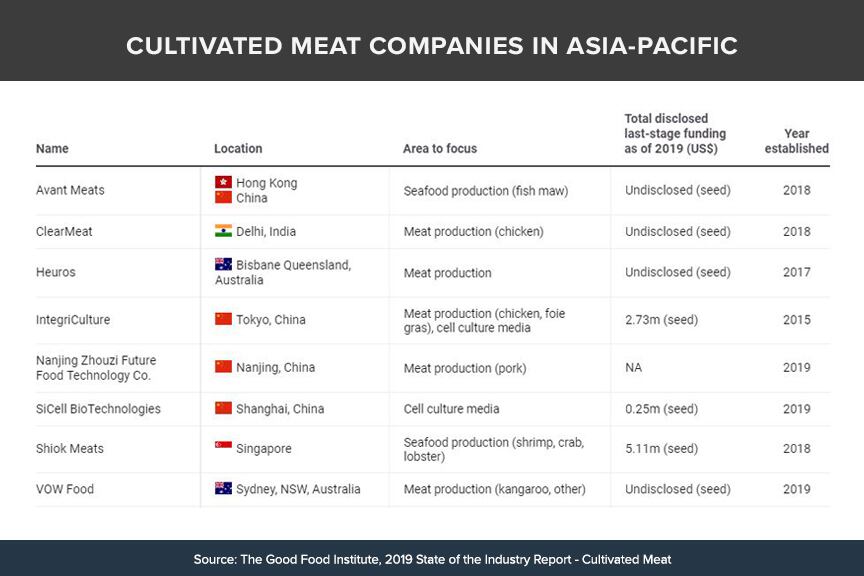
As of end-2019, 55 cell-based meat and seafood start-ups have been established globally, of which just eight come from the Asia Pacific region, according to data from the Good Food Institute’s Cultivated Meat Industry report.
These were: Australia’s VOW Food (kangaroo meat) and Heuros (meat), Singapore’s Shiok Meats (seafood), China’s SiCell BioTechnologies (cell culture media) and Nanjing Zhouzi Future Food Technology Co (pork), India’s ClearMeat (chicken), Japan’s IntegriCulture (chicken, foie gras, cell culture media), and Hong Kong’s Avant Meats (seafood).
GFI uses ‘cultivated meat’ in place of ‘cell-based meat’, although the terms are generally recognised to be interchangeable along with ‘clean meat’ and ‘cultured meat’.
Despite taking up just a small fraction of the world’s cell-based meat start-ups, countries in the APAC region are expected to take the lead when it comes to developing regulatory governance for the sector, according to GFI APAC Managing Director Elaine Siu.
“My personal feeling is that the first cultivated meat product is probably going to be approved in Singapore due to food security concerns and the government is also hoping to get these products out to market,” she told FoodNavigator-Asia.
“The biggest challenge when it comes to cultivated meat regulations is that it’s something of a chicken-and-egg issue: The regulators need to see what products start-ups are coming up with so they can create a structure to actually assess these products, whereas start-ups are looking to the regulators to see what they are concerned about so their products can be approved.
“In places like Singapore where the government is committed to facilitate the growth of this sector, then it makes things much easier as the regulator is actually proactive in getting conversations going and understanding what is being developed.
“So it really actually has to be a parallel development for both products and regulations [for this to work out best].”
She added that in Japan, cultivated meat governance is currently not expected to take the same route as most other countries which consider this under the ‘novel food’ category.
“Theoretically, cultivated meat products in Japan can enter the market today – no new regulations need to be created, [although] industry groups are working to create industry standards. That is really very exciting, as the law is not even in the way,” she said.
“As it is, I feel that we might actually see Asia leading the regulatory development in this space as compared to other countries such as the United States and Europe – as mentioned, Japan has already removed a huge hurdle by not requiring new laws to be instated, and Singapore is really committed to bringing these products to market as soon as possible.”
Watch the video below for more of her insights:
Japan and Singapore running ahead
Japanese cell-based meat firm IntegriCulture is likely the start-up mentioned by Siu in the interview above as it produces its own cell culture media which does not contain growth factors.
The firm’s CEO Yuki Hanyu confirmed with us that ‘cell-based meat is not novel’ in Japan.
“Existing food regulations would apply to cell-based meats here, [so no new regulations are needed] although new regulations may be introduced if significant consumer actions are taken or health and safety incidents are reported,” he said.
In Singapore on the other hand, new regulations are still required for this sector under the novel foods category, and the development of these falls under the purview of the Singapore Food Agency (SFA), for which food safety is a major concern for investigation before formally launching any regulations.
“As novel foods do not have a history of being consumed by humans as food, they may pose food safety risks and their safety needs to be assessed before they can be allowed to be used in food for sale in Singapore,” SFA told FoodNavigator-Asia in a statement.
“[Currently, companies are required] to seek pre-market approval for novel foods that do not have a history of use as food. [These will need to undergo a] novel food safety assessment that covers food safety issues including potential toxicity, allergenicity, safety of its production method, and dietary exposure.
“Novel food products will be assessed on a case-by-case basis before import or sale is allowed. Companies that wish to import or manufacture novel food products for sale are required to seek SFA’s approval and provide the necessary supportive evidence of food safety for our assessment.”
India and China catching up
Singapore cell-based seafood start-up Shiok Meats’ CEO Sandhya Sriram concurred with Siu that Singapore is likely the front-runner in terms of regulatory landscape, but also highlighted India as a possible upcoming contender here.
“In India, this would fall under the purview of the Food Safety and Standards Authority India (FSSAI). GFI India has organised meetings about this, and I was at the first one and saw they are also combing the landscape and at least actively looking to find out more about it,” she told us previously.
“I’m glad that one of the biggest countries in Asia is at least starting to recognise this and think about it, which is really more than enough to get things moving at this point.”
China is also looking at developing its cell-based meat sector, with calls for a national strategy focusing on this area raised during a recent high-level governmental meeting.
“Cell-based meat is being viewed as the most likely solution to meat supply via clean and sustainable means,” said National Committee of the Chinese People's Political Consultative Conference (CPPCC) member Sun Baoguo.
“Cell-based meat analysis and technological advancements are a key way to guaranteeing China’s future meat supply, and gaining a leading position when it comes to this production technology will also have important strategic importance for the country.”
Australia’s Vow, which is looking at the development of more unique cell-based meats such as kangaroo, agreed that Singapore is making big strides in this area, but Australia is working on this too.
“We have made a preliminary submission to Food Standards Australia New Zealand (FSANZ) and are working with them on this,” Vow Co-Founder and Chief Commercial Officer Tim Noakesmith told us.
“They are actively looking to learn more about the process and outcomes, and we are working closely together on bringing products to market. FSANZ places a lot of importance on a high quality regulatory system, which is also important so as to preserve the quality of the Australian brand.”
Outside of APAC
Israel is commonly acknowledged as one of the world’s cell-based meat leaders with many key start-ups in the sector based there.
One of these is Aleph Farms, which produces cell-based beef, and also believes that regulatory guidance is needed to develop an effective framework for the sector.
“There is a need for public–private collaboration, a proactive dialogue with regulators and integration of suitable products into local food cultures, history, and cuisines,” Aleph Farms’ Co-founder and CEO Didier Tobia told us.
“National regulatory agencies are looking to align with the global–local approach, [and] benchmark either US or European [cell-based meat regulatory] procedures.”
As Israel is primarily a Jewish nation, another primary concern for the local sector is whether or not cell-based meat is considered kosher, and issue currently under debate.
“There is a debate on which type of kosher label clean meat will fall under; in Judaism, any food needs to be labelled as meat, dairy or neither, which we call parev, due to kosher laws that prevent Jews from having dairy and meat at the same meal,” Israeli-based cell-based meat firm SuperMeat Co-Founder and Chief Commercial Officer Shir Friedman told Culture Trip.
“If clean meat is considered parev, that could actually open the door for a kosher cheeseburger, a delight that Jews are prevented from having.”

Meanwhile in the EU, current food laws have added a possible extra complication for producers with a nutritional angle.
“In the EU, the law requires that any ‘novel’ food must offer the same nutritional values as an equivalent already on the market,” Dutch cell-based meat firm Mosa Meats’ spokeswoman Beckie Calder-Flynn told FoodNavigator-Asia.
Mosa Meats’ Chief Scientific Officer Professor Mark Post is known as the ‘founding father of cultured meat’ as he created the world’s first first cell-based beef hamburger back in 2013.
“Because our meat is taken in biopsies from cows, this is not an issue for us as opposed to if someone was creating the meat from scratch. It is also worth noting that our meat is not genetically modified, so this will not be an additional part of the regulation process,” said Calder-Flynn.
That said, this adds a level of complication in terms of firms in the EU having to solidly prove that cell-based meat products provide the same level of nutrition as regular slaughter-based meat in every way, which could be easier said than done depending on the level of evidence required.
How long till affordable products?
The expectations of each firm in terms of reaching price parity, or at least affordability by the masses, currently varies widely between countries and depending on product type.
Aleph Farms has just opened a pilot plant dubbed BioFarm to scale up production of its cultivated beef products, which it expects to accelerate price parity for its meat.
“Same as with any new food product that reaches the market, our products at the beginning going to be slightly more expensive than slaughter-based meat. With the building of large-scale BioFarms, the price will decrease and reach parity with slaughter-based meat within a few years,” Toubia said.

Shiok Meats will probably be one of the first to hit affordable pricing based on current estimates.
“We are aiming to reduce the price of our cell-based shrimp products to US$50 per kg by the end of 2020, and officially launch in restaurants in Singapore as early as 2021,” said Sriram.
The usual market price for shrimps on the global market is around US$14 to US$15, although this has dropped in recent months due to COVID-19 – but the firm also hopes to match regular shrimp market prices eventually.
One of the major barriers faced by these firms when it comes to bringing down price is in the cost of growth media, which Japan’s Integriculture has tried to address by developing its own cell culture media without growth factors.
In place of growth factors, which are normally used to help the muscle cells (most commonly consumed portion) grow, the company cultures a variety of in separate bioreactors using a closed loop system.
“Through this whole body emulation, we removed the need to add serum or growth factors, breaking the ‘priciness’ barrier,” said Hanyu.
That said, despite having removed growth factors, the firm still faces a major hurdle in bringing down the cost of its yeast and algae culture medium – it aims to reach a cost between US$0.01 to US$0.5/L by 2025 instead of the current US$1 to US$10/L.
IntegriCulture is currently building and scaling its production line for cell-cultured foie gras, scheduled to operate by December 2021.
Timelines and investments
Based on all of this information, Siu’s prediction that Singapore could be the first to formulate cell-based meat regulations may not be far off, and this could happen in as soon as 12 months.
“I would really expect to hear news from Singapore on these cultivated meat regulations in the next year,” she said.
“They’re already taking in and accepting applications and developing safety protocols as it is, [so it should be soon].
“When products are ready to actually go to market based on cost and scalability in the different markets is another timeline altogether, but I would say the regulations side of things is definitely not going to be the one [slowing down the development of the sector].”
Toubia also expects fast regulatory progress in Israel given the current rate of cell-based development in the country, but still not as fast as in Singapore.
“[I would say that] 2022 could to be the year where regulatory approval on the safety and quality of products and large-scale cultivation facility will occur [here],” he said.

Despite nothing being set in stone yet in terms of regulations, cell-based product start-ups are still obtaining large amounts of funding and investment – case in point Shiok Meats’ recent US$3mn bridge funding, and also Turtletree Labs which produces cell-based milk.
The latter recently raised US$3.2mn in its seed round and won the Temasek Foundation’s Liveability Challenge to obtain an additional SGD1mn (US$719.3mn).
“When we first started out in 2019, we were at US$180/L using costly pharma grade components. Recently, we have synthesised our own proprietary lactation media that has enabled us to drive costs down greatly,” Fengru Lin, TurtleTree Labs’s founder and CEO told us.
“There is an advent of cell-based food companies in the region as compared to two years ago and I believe that with a growing ecosystem, this would accelerate the development of the region’s regulatory landscape around novel food.”
The firm is building a 5L bioreactor to be completed by end-2020, as well as designing a full-scale pilot plant, which these funds will go to.
As for why so much money is still available for start-ups in a sector that has not yet actually found stable ground in terms of regulations and production despite the risk involved, Siu told us that this was due to the ‘certainty’ of the need for the sector.
“Start-ups in the sector are able to secure large investments even though nothing is as yet certain [with regard to product development and governance], because it is certain that the current animal-based food system does not work,” she said.
“The COVID-19 pandemic has made it much more certain in everyone’s minds that what we have currently is inefficient, [and] cultivated meat is one of the solutions that can help to fulfil part of the required [protein] supply.”




