The paper tested 35 samples of Ginkgo biloba food supplements bought from health food stores, supermarkets and pharmacies in London and on the internet.
Using nuclear magnetic resonance (NMR) spectroscopy they found the vast majority were not up to quality or labelling scratch.
Most contained higher levels of the flavonols rutin and/or quercetin or lower levels of ginkgo metabolites compared to reference samples of the leaves.
“Adulteration of ginkgo products sold as unregistered supplements within the very large market of ginkgo products (reputedly £650 million annually) through the post-extraction addition of cheaper (e.g. buckwheat derived) rutin is suspected to allow sub-standard products to appear satisfactory to third parties, e.g. secondary buyers along the value chain or any regulatory authorities,” the researchers wrote in the Journal of Herbal Medicine.
“This study was therefore carried out to identify products that did not conform to their label specification and may have been actively adulterated to enable access to the global markets.”
The paper's authors from the UCL School of Pharmacy, University of Birmingham School of Biosciences and University of Westminster in the UK along with Swiss NMR spectroscopy specialist CAMAG, warned consumers were likely to assume an unregistered ginkgo food supplement contained the same extract as a registered herbal medicine, which their results revealed was not always the case.
Ginkgo – often marketed as improving circulation, memory and cognitive function – is a licensed drug in some European countries like Germany.
In the UK, two ginkgo products hold a Traditional Herbal Registration (THR) for the treatment of Raynaud’s disease – a condition affecting circulation – although the majority of products on the UK market are sold as 'unlicensed' over-the-counter botanical food supplements.
The European Traditional Herbal Medicinal Products Directive (THMPD) – active since May 2011 – requires each EU member state to set up a traditional herbal registration scheme for manufactured traditional herbal medicines suitable for use without medical supervision.
In the UK manufacturers should have the appropriate product license from the Medicines & Healthcare Products Regulatory Agency (MHRA) to legally sell herbal medicines.

To apply for Traditional Herbal Registration (THR) - which only covers medicine used for minor health conditions where medical supervision is not required like colds - a company must submit:
• a technical dossier covering the quality of herbal ingredients
• a review of safety with an expert report including clinical and non-clinical safety areas carried out by either a registered doctor, pharmacist, a herbal practitioner who is a member of a professional body or a scientifically qualified individual like a toxicologist
• a draft summary of product characteristics according to the European Commission’s guidelines
• a mock up label and patient information leaflet, according to the best practice guidelines
Companies with products targeting more serious conditions must seek a Marketing Authorisation (MA) license.
Fingerprinting
From a regulatory perspective, five of the 35 samples were not compliant with label specification.
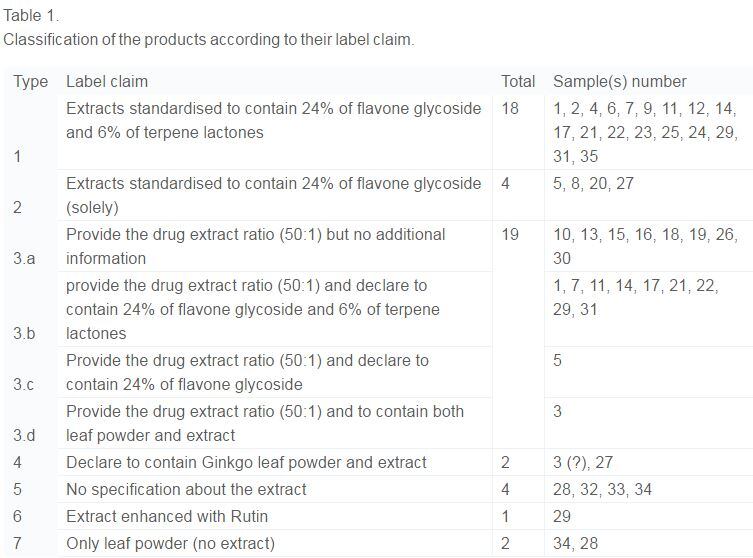
The other 28 samples were in line with their label claims but quality was different from that outlined in pharmacopoeias, and their ‘fingerprints’ differed in terms of the phenolic compounds rutin and quercetin.
One product even contained a 5-hydroxytryptophan derivative, which is not a natural constituent of ginkgo.
Only two samples had ‘fingerprints’ similar to pharmacopoeia monographs for ginkgo extract.
So what’s the solution?
The researchers urged all producers of ginkgo final products to gain a “detailed understanding of the value chains of their products”.
They also said NMR spectroscopy and CAMAG’s thin layer chromatography (TLC) system were useful tools in providing “further evidence as to the degree and kind of adulteration of gingko supplements”.
Yet CEO of the European arm of Chinese firm Beijing Gingko Group (BGG) Christian Artaria told us: “What is stated in this paper is something known by the industry. As an industry expert, knowing the existing analytical tools in the industry, using an NMR to underline this issue sounds to me a little bit too much: like using an atomic bomb to kill a mouse when a mousetrap could have done the work, of course always if you want to place and check your mousetraps!”
NMR spectroscopy uses the magnetic properties of atomic nuclei to provide detailed information about the structure and dynamics of molecules.
Artaria said methods like TLC and High Performance Liquid Chromatography (HPLC), which he said he hoped most companies were already using, were sufficient.
Spot the price difference
The ex-global marketing chief of Italian botanical firm Indena told us: “Unfortunately the reality is that people don't want to see what is an evident issue when they purchase ginkgo extracts at incredible prices!”
Back in 2013 founder and executive director of the American Herbal Pharmacopoeia (AHP) Roy Upton told our sister publication NutraIngredients-US he believed the issue of ginkgo adulteration was “very widespread”, with a 30-50% cost difference between suppliers.
Last year the Chinese Food and Drug Administration (CFDA) recalled an estimated 200 million ginkgo pills in an attempt to crack down on companies supplying substandard versions of the botanical.
According to a paper from the EU-funded PlantLIBRA project, one in five Europeans take plant food supplements with ginkgo being one of the most popular.
Source: Journal of Herbal Medicine
Published online ahead of print, doi:10.1016/j.hermed.2016.04.003
“Adulteration and Poor Quality of Ginkgo biloba Supplements”
Authors: A. Booker, D. Frommenwiler, E. Reich, S. Horsfield and M. Heinrich
