Korean player Daesang moves on EU omega-3 market

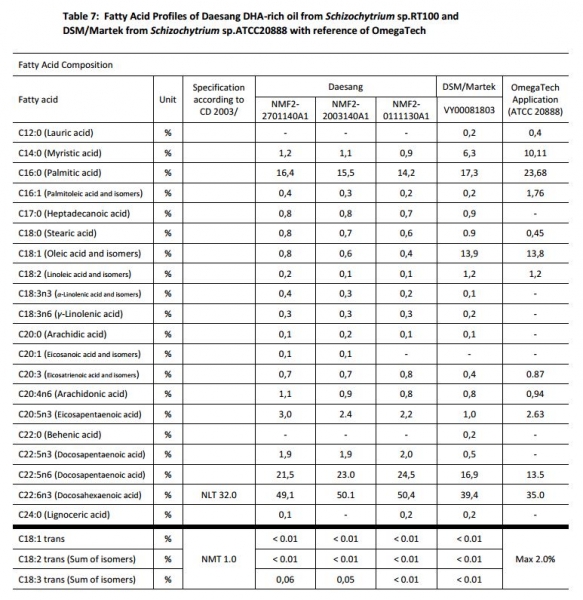
The application seeks ‘substantial equivalence’ with DSM-Martek’s Schizochytrium-derived DHA that was approved for use in the European Union food supply in 2003.
“Taken together, given the fact that the DHA content of Daesang DHA-rich oil is in compliance with the authorised product specifications (CID 2014/463/EU) and given the relatively minor differences in percentage for some of the other fatty acids, we consider the fatty acid profiles of our DHA-rich oil to be substantially equivalent to DSM/Martek DHA-rich oil,” the firm said in its application to the UK Food Standards Agency (FSA).
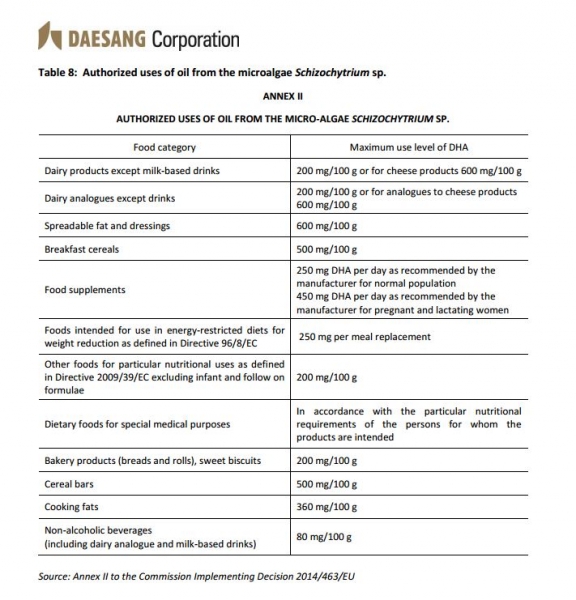
Schizochytrium-derived DHA is approved for use in a wide variety of formats across the EU including dairy products, cereals, food supplements and medical foods.
Daesang manufactures a range of foodstuffs including margarine, coffee and pasta and has a suite of ingredients including starch, chrolella and algae.
There are nine DHA-related approved health claims in the European Union relating to brain health, heart health and vision in adults and infants and foetuses. Some of the claims are in conjunction with omega-3 form EPA (eicosapentaenoic acid).