China direct
Economic slowdown now squeezing margins in China's agri-sectors

This is according to Rabobank in a new report that looks at how businesses have been weathering stock market volatility and currency shifts in the country. In it, the agri-business specialist bank says higher input costs have been driving F&A companies to address their cash flows.
Market volatility means they also face difficulties raising stock market funds and managing currency exposures. The report predicts this may prompt China’s F&A companies to review their offshore sourcing options for commodities and products, as well as M&A plans. The greatest impacts are forecasted for F&A companies importing raw materials.
“Chinese food producers and manufacturers will need to adjust their production and product approaches to achieve strong margins,” said Sandy Chen, an F&A analyst for the bank.
“We forecast the dairy processing industry could be one of those most affected, where higher input costs as a result of a substantial depreciation could deflate pre-tax profits by 15%.”
Chenjun Pan, also an analyst at Rabobank, added: “Raw beef imports, which make up 12% of the domestic market, may also become increasingly unaffordable in the face of tight global supply. This will put upward pressure on China’s beef prices, which will be difficult to pass on to consumers in the weaker economy.
“While it will be increasingly difficult for China’s F&A companies to raise funds, the flow-on financial turbulence in other markets may make some overseas beef assets, more affordable for financially strong Chinese companies.
“Owing to the country’s long-term beef shortage, Chinese companies will remain strongly interested in overseas growth opportunities.”
Rabobank also expects the devalued yuan and slowing growth the hit the brewery sector, which relies on barley imports for producing beer, and the country’s instant noodle and bakery industries, which depend heavily on imported palm oil for noodle processing and wheat flour for baked goods.
The bank also predicts that Chinese F&A companies may become more attractive for foreign investment with businesses in the farm inputs and Chinese spirits sectors cited as potentially attractive investments.
US supplements import group queries some of China’s draft regulations
The US–China Health Products Association (USCHPA) has sent China’s food regulator its response to a draft regulatory framework to govern the dietary supplement industry.
The USHPA’s submission to the China Food and Drug Administration (CFDA) can be viewed at the association’s website.
Jeff Crowther, the USHPA’s executive director, said he was happy to see China reforming its supplement regulations, which have been especially chaotic and extremely difficult to follow for all involved.
The two most significant changes for the new framework will be the creation of a product filing system and an approved raw ingredient catalogue.
The filing system is expected to be akin to a type of notification system, though only vitamins, minerals and some other select nutrients will be allowed to make use of it in a bid to help increase transparency and speed entry to market.
However, Crowther says indications suggest that products containing complex formulas, especially those containing botanicals, will still have to go through the current registration process, which typically costs in excess of US$80,000 per product unit and can take up to three years to finalise.
“USCHPA is hoping this registration process will also see some streamlining and much needed transparency,” he said.
The approved raw material catalog will be created by CFDA in conjunction with the National Health and Family Planning Commission. This list will help the industry to understand better the ingredients they can use when formulating finished products.
Of the CFDA’s draft, USCHPA has questioned the need for imported products to have a record of sales and marketing that goes back at least one year in the country of origin prior to attempting entry. It also queried by they show the results of three consecutive batch records.
“This is not obviously required for domestic companies and is a form of non-tariff barrier to trade,” said Crowther.
The association also took issue with wording that states that imported products are expected to follow China’s quality standards and that on-site inspections would not be out of the question.
“This wording needs clarification, but could require foreign manufacturers to follow Chinese GMP standards for health food products, which would certainly be problematic,” said Crowther.
Woman arrested for selling slimming product containing banned drugs
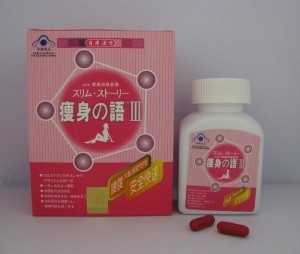
A 24-year-old Hong Kong woman has been arrested in an operation involving the police and the health department for the suspected illegal sale of a slimming product that was suspected to contain undeclared banned drug ingredients.
Following a consumer complaint, it was found that a local internet seller had been offering for sale the slimming product (pictured), whose name does not have an English translation.
A sample of the product was purchased for analysis and test results from the government laboratory showed the product contained the banned pharmaceutical ingredients sibutramine and phenolphthalein.
During their sting, the police arrested the seller for suspected illegal sale of an unregistered pharmaceutical product and a Part I poison.
Sibutramine is classed as a Part I poison when it is used as an appetite suppressant. Since November 2010, products containing sibutramine have been banned because of increased cardiovascular risk.
Phenolphthalein was once used to treat constipation, but has since been banned in Hong Kong for its cancer-causing effects.
According to the Pharmacy and Poisons Ordinance, all pharmaceutical products must be registered with the Pharmacy and Poisons Board of Hong Kong before they can be legally sold in the market.
Part I poisons can only be sold in a pharmacy under the supervision of a registered pharmacist. Illegal sale of these and other unregistered pharmaceutical products are criminal offences with a maximum penalty of a HK$100,000 (US$13,000) and two years' imprisonment.
"Weight control should be achieved through a balanced diet and appropriate exercise. The public should consult healthcare professionals before using any medication for weight control," a health ministry said.